You can measure the weight of your sample using an analytical balance (also called a “lab balance”), a device that weighs substances in grams. 
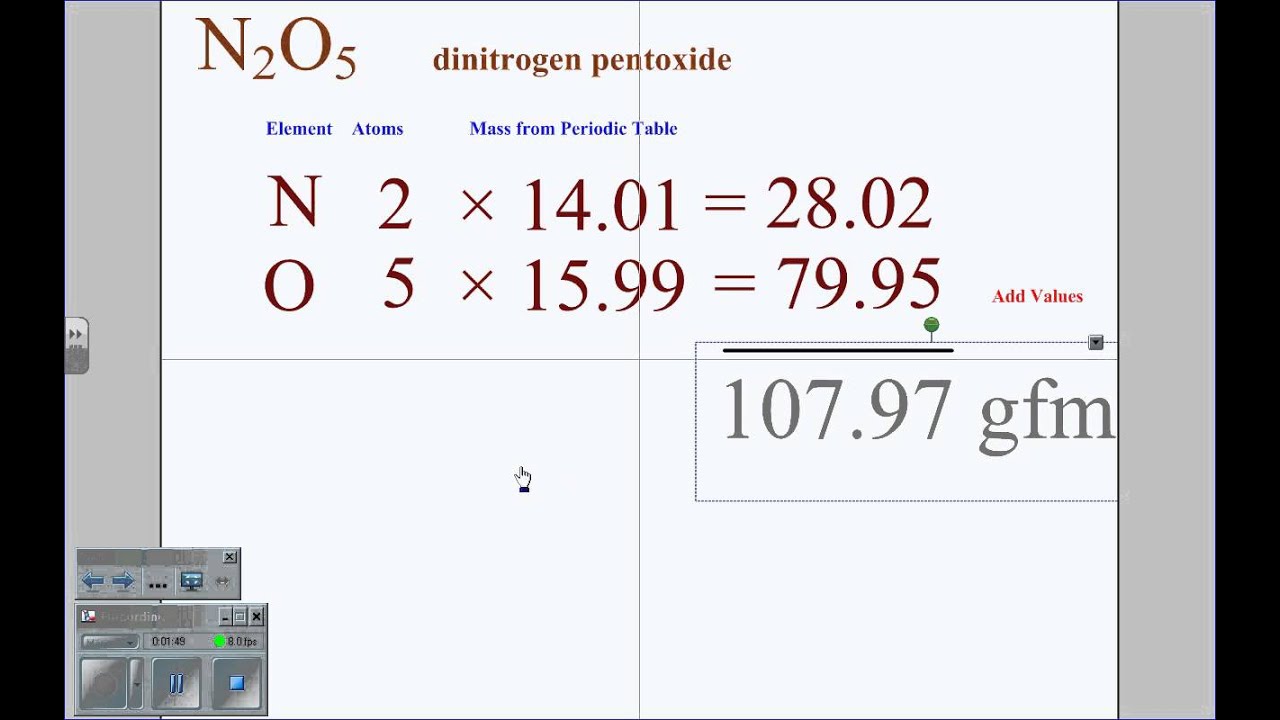
Next, you need to find the ‘physical’ weight of the compound in question, which, in this case, is sodium chloride. This is how you can determine the molecular mass of any compound. 
Thus, the molecular mass of NaCl turns out to be 58.52 grams/mol. Molecular mass of NaCl = atomic mass of sodium + atomic mass of chlorine The atomic mass of sodium is 22.98, while the atomic mass of chlorine is 35.543. NaCl is made of two atoms: sodium (Na) and chloride (Cl). Let’s consider the example of NaCl (sodium chloride or common salt) to understand this concept better. A compound is composed of a number of atoms of different elements, and their combined weight becomes the molecular weight of the compound. The first step is to determine the molecular weight (or molecular mass) of the compound you’re dealing with. Now that we understand what a mole is, it’s time to proceed and look at how to determine the number of moles present in a chemical compound, provided that we already know the physical weight of the compound in question. No matter what their molecular masses are, all of them contain the same number of molecules for a particular number of moles, i.e., 1 mole of glucose contains the same number of molecules as 1 mole of methane. The same applies in the case of chemical compounds. The respective masses of hydrogen and oxygen, however, are different. For instance, 1 mole of hydrogen and 1 mole of oxygen both contain approximately 6.022 x 10 23 atoms. Here’s an interesting thing about moles: 1 mole of any element or chemical is always the same number. Just as it’s much simpler and more convenient to measure intergalactic distances in light-years instead of centimeters, it’s easier to count atoms in moles rather than counting them in billions and trillions. When chemists perform a chemical reaction in a laboratory, they want to ensure that they are using the right amounts of all chemicals involved in the reaction before actually starting the process. While the former is a unit of measurement, the latter is a dark spot on the skin comprised of skin cells that have grown in a group. Note that the mole in chemistry should not be confused with the mole related to the human body. In short, you could say that a “mole” is the unit of amount in chemistry, i.e., it tells you how much of a particular chemical is present. have measurement units (meters, grams, seconds respectively), similarly, chemicals are measured in moles. Just like how physical quantities, such as distance, mass, time etc. Moles are a unit of measurement of chemical compounds. I also want to state an easier, simpler-to-understand definition of a mole (of chemistry). Carbon-12 is a Carbon isotope whose nucleus contains six protons and six neutrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2023
Categories |
 RSS Feed
RSS Feed
